For up to date information on the TRACK-TBI, please visit the TRACK-TBI NETWORK website:
*****
Traumatic Brain Injury (TBI) is a condition, not a one-time event. Effective treatment of TBI represents a great unmet need in public health. In 2013, approximately 2.8 million TBI-related emergency department (ED) visits, hospitalizations, and deaths occurred in the United States. TBI is a contributing factor in a third of all injury-related US deaths. An estimated 3.2 to 5.3 million people live with the long-term physical, cognitive, and psychological health disabilities of TBI, with annual direct and indirect costs estimated at over $76 billion. Although we are gaining ground in our understanding of the pathophysiology of TBI, these advances have failed to translate into a single successful clinical trial or treatment.
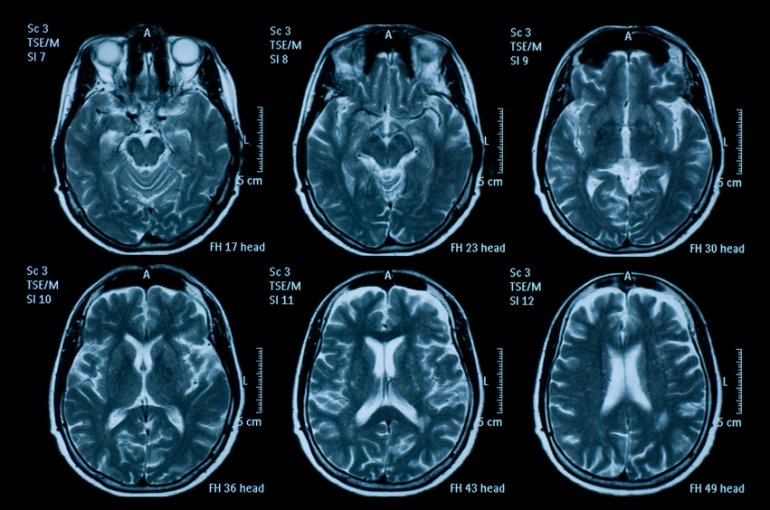
The NINDS-funded, multicenter Transforming Research and Clinical Knowledge in Traumatic Brain Injury (TRACK-TBI) study aims to change this. In collaboration with expert public-private partners, we have reached our enrollment goal of 3,000, having collected and analyzed detailed clinical data on subjects at 18 U.S. sites, across the injury spectrum, along with CT/MRI imaging, blood biospecimens, and detailed clinical outcomes. This rich and diverse precision medicine dataset will reside in a publicly accessible infrastructure of integrated databases, imaging repositories, and biosample repositories – a true TBI Information Commons. Our high quality and fully integrated TBI-Common Data Element dataset will provide a global platform to connect TBI’s best scientists.
TRACK-TBI is built from the foundational NINDS-funded TRACK-TBI Pilot study, which collected clinical data from 3 sites and 600 subjects. With over a dozen publications and growing, the TRACK-TBI Pilot dataset is the first to populate the Federal Interagency Traumatic Brain Injury Research (FITBIR) repository and with the current TRACK-TBI data, is compatible with the International Initiative for Traumatic Brain Injury Research (InTBIR), a collaborative effort of the European Commission (EC), the Canadian Institutes of Health Research (CIHR) and the National Institutes of Health (NIH).
TRACK-TBI also forms one of the core datasets of the Department of Defense-funded TBI Endpoints Development (TED) Initiative, which has established an interrogatable Metadataset of high quality studies across civilian, sports, and military populations. TED’s goals, aligned with TRACK-TBI, are to identify and validate the FDA regulatory readiness of candidate clinical outcome assessments and proteomic and neuroimaging biomarkers and technologies that may serve as endpoints in the design of precision clinical trials.
The TRACK-TBI fully integrated TBI Information Commons will:
- Improve TBI classification/taxonomy for targeted clinical treatment trials
- Improve TBI outcome assessments, such that the size and costs of clinical trials can be reduced
- Identify the health and economic impact of Mild TBI patient disposition
- Create a legacy database with analytic tools and resources to support TBI research
Our expected outcome for this international resource is to identify new diagnostic and prognostic markers and refine outcome assessments, which will lead to successful clinical treatment trials.